FEATURE
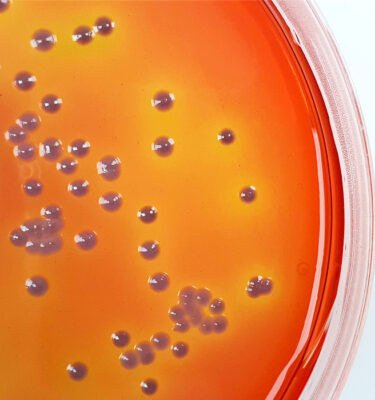
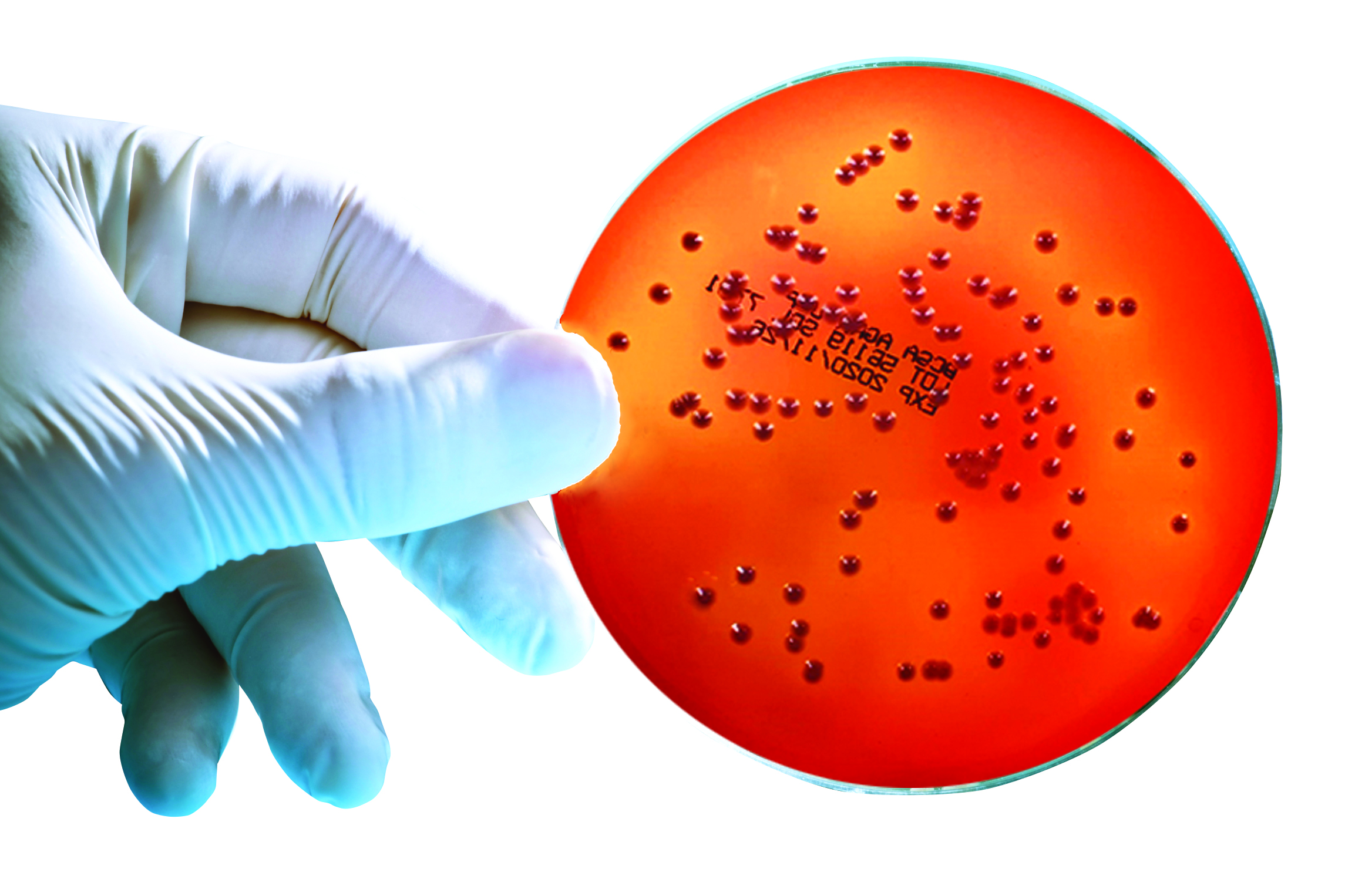
SGL Burkholderia cepacia Selective Agar (BCSA) in 90mm plate format is a selective growth medium specifically designed to meet the formulation requirements listed in USP <60> to recover Burkholderia cepacia, a bacterial species of rising importance in the pharmaceutical, clinical and cosmetic industries in recent years.
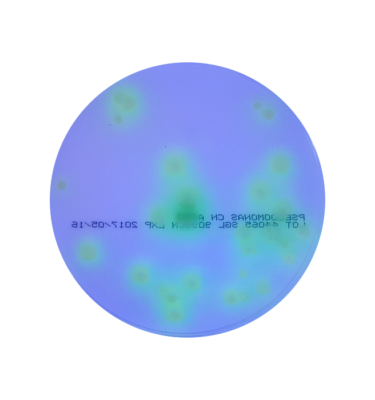
Burkholderia cepacia is a member of a group of more than 20 closely related species in the Burkholderia cepacia complex (BCC). BCC strains are some of the most versatile Gram-negative bacteria with a wide environmental distribution in soil, plants, and water.
Impact of Burkholderia cepacia on pharmaceuticals
BCC is one of the contaminants most frequently encountered in sterile and non-sterile pharmaceutical products.
Health–care associated outbreak of BCC have been linked to contaminated nebulized and intravenous solution, including commercial distributed products, skin care product and disinfectant.
In the past decade, organisms that fall within the BCC grouping have been identified as potentially ‘objectionable’ within the pharmaceutical manufacturing environment
Testing for Burkholderia cepacia
Until recently there was no compendial testing procedure detecting BCC in pharmaceutical components or finished product.
On December 1, 2019 The United States Pharmacopoeia (USP) published chapter <60> Microbiological Examination of Non-sterile Products – Tests for BCC. The USP <60> specifies the test strains for growth promotion, suitability methods and selective media recommended to ensure the absence of BCC.
With the creation of the USP<60>, the pharmaceutical industry has taken a definitive step towards addressing the public health risks posed by B. cepacia.
Why use SGL Pre-poured BCSA?
- Meets the USP formulation criteria
- Quicker and better isolation of BCC
- Increased selectivity
- Fewer false positive
- Saving laboratory time
- Consistent media every time
SGL BCSA pre-prepared media contains numerous nutritional components, and energy sources that support BCC growth, and a mixture of antibiotics designed to prevent the growth of non-fermenters not belonging to the BCC and typical environmental isolates such as Pseudomonas and Staphylococcus species.
This allows for easier detection of BCC in test samples containing background contaminants.
All these elements make BCSA more selective and efficient in growing BCC colonies than other media such as Burkholderia cepacia agar (BCA), and Oxidation-Fermentation Polymyxin Bacitracin Lactose agar (OFPBL).
SGL supplies a wide range of other pre-poured media for use in pharmaceutical, cosmetic, clinical and veterinary applications. These include standard off the shelf media as well as bespoke media products tailored to customer specific formulations.
Representative samples of our products are QC tested in our on-site laboratory which is UKAS accredited to ISO 17025.
View All Pharmaceutical Application Products
Other Products for Pharmaceutical Applications
-
iVAS Roam Portable Microbial Air Sampler
Read more -
Tryptone Soya Broth, Bag with Spike Port & Capped Needle-Free Port
Read more -
Tryptone Soya Broth Bag with Needle-Free Port
Read more -
Stericheck™ OVP- C
Read more -
Burkholderia Cepacia Selective Agar (BCSA), USP
Read more -
Fluid D USP – Universal Vial
Read more -
Phosphate Buffer Solution, Universal
Read more -
Sabouraud Dextrose Broth, (EP, USP), DIN bottle
Read more -
10% Glycerol Nutrient Broth, Syrup
Read more -
Buffered Peptone Water + 5% Polysorbate 80 (Tween 80), Syrup
Read more -
Thioglycollate Medium (USP), Bag
Read more -
Eugon LT100 Broth, Universal
Read more -
Buffered Peptone Water + 5% Polysorbate 80 (Tween 80), Syrup + Beads
Read more -
Buffered Sodium Chloride Peptone Broth, Harmonised (EP, USP, JP), Tube
Read more -
EST Neutraliser + Saponin, Universal
Read more -
Tryptone Soya Broth (TSB) EP, USP, Syrup (Sterile Outer Bagged)
Read more -
Dey/Engley (D/E) Neutralising Broth, Syrup
Read more -
Antibiotic Medium No. 8, Syrup
Read more -
Antibiotic Medium No. 32, Winchester
Read more -
Antibiotic Medium No. 32, bottle
Read more -
Reinforced Clostridial Medium (RCM), Syrup
Read more -
0.0375% Tetrazolium Chloride, bottle
Read more -
Saline – Physiological (0.9%), Bagged
Read more -
Thioglycollate Medium (EP, USP), Tube
Read more -
TSA (Tryptone Soya Agar), Quad wrapped 140mm Plate, Irradiated
Read more -
TSA Aircheck® 4W (Tryptone Soya Agar), 90mm Plate, Irradiated
Read more -
Quick ViewApplication, Bacteria & Fungi, Bio Burden, By Product Group, By Sector, Clinical & Veterinary, Contact Plate, Environmental Monitoring, Feature Page, General Purpose, Irradiated plates, Irradiated Plates, Organism, Pharma, Pharmaceutical & Cosmetic, Pre-Poured Plates, Surface Monitoring, Type, Water & Environmental
TSA Surfacecheck® N 4W (Tryptone Soya Agar+Neutralisers), Quad wrapped Contact Plate, Irradiated
Read more -
SDA Surfacecheck® N 4W (Sabouraud Dextrose Agar + Neutralisers), Quad wrapped Contact Plate, Irradiated
Read more -
SDA Aircheck® 4W (Sabouraud Dextrose Agar), Quad wrapped 90mm Plate, Irradiated
Read more -
Buffered Peptone Water, Bag
Read more -
Stericheck™ OVP-B
Read more -
Stericheck™ OVP-A
Read more -
Phosphate Buffered Saline (PBS), Bijou
Read more -
MacConkey Broth EP/USP, Syrup + Durham Tube
Read more -
Quick ViewApplication, Bacteria & Fungi, Bio Burden, By Product Group, By Sector, Clinical & Veterinary, Contact Plate, Environmental Monitoring, Feature Page, Featured, General Purpose, Irradiated plates, Irradiated Plates, Organism, Pharma, Pharmaceutical & Cosmetic, Pre-Poured Plates, Surface Monitoring, Type, Water & Environmental
TSA Surfacecheck® (Tryptone Soya Agar), Contact Plate, Irradiated
Read more -
Pseudomonas CN Agar, 90mm Plate
Read more -
Gram’s Stain — Crystal Violet, Concentrate, Syrup
Read more -
Gram’s Stain — Lugols Iodine, Concentrate (Sufficient for 250ml), Universal
Read more -
Neutral Red, Concentrate, Syrup
Read more -
Safranin, Concentrate, Syrup
Read more -
Colmubia CNA Agar, 90mm Plate
Read more -
Columbia Blood Agar + 0.5% Yeast Extract + 5% Defibrinated Horse Blood, 90mm Plate
Read more -
Desoxycholate Citrate Agar (DCA) Hynes, 90mm Plate
Read more -
Desoxycholate Citrate Lactose Sucrose Agar (DCLS), 90mm Plate
Read more -
MacConkey Agar with Salt, 90mm Plate
Read more -
MacConkey Agar No. 3 (+ Salt and Crystal Violet), 90mm Plate
Read more -
Nutrient Agar + Salt, 90mm Plate
Read more -
Sabouraud Dextrose Agar + Chloramphenicol, 90mm Plate
Read more -
Tryptone Soya Agar + 5% Horse Blood, 90mm Plate
Read more -
Tryptone Soya Agar + 5% Sheep Blood, 90mm Plate
Read more -
Lactose peptone water + Phenol Red, Bijou
Read more -
Aesculin Bile Azide Agar, 90mm Plate
Read more -
Brilliant Green Agar (Modified) + Novobiocin, 90mm Plate
Read more -
Rose Bengal Chloramphenicol Agar, Syrup
Read more -
Perfringens TSC Agar, 90mm Plate
Read more -
Pseudomonas CFC Agar, 90mm Plate
Read more -
Brilliant Green Bile Broth with Durham tube Double Strength, Universal
Read more -
Brilliant Green Bile Broth (+ Durhams Tube), Universal
Read more -
Tryptone Water, Bijou
Read more -
Tryptone Water, Universal
Read more -
Maximum Recovery Diluent (MRD), Honey Jar
Read more -
Maximum Recovery Diluent (MRD)
Read more -
Maximum Recovery Diluent (MRD)
Read more -
Maximum Recovery Diluent (MRD)
Read more -
Plate Count Agar (PCA), Syrup
Read more -
Plate Count Agar (PCA), Winchester
Read more -
Defibrinated Horse Blood, Bottle
Read more -
Lysed Horse Blood, Bottle
Read more -
Defibrinated Sheep Blood, Bottle
Read more -
D/E Neutralising Broth, Syrup
Read more -
D/E Neutralising Broth, Universal
Read more -
Dey/Engley (D/E) Neutralising Broth + 2% Tween 80, Syrup
Read more -
Horse Serum, Bottle
Read more -
Buffered Peptone Water, Honey Jar
Read more -
Buffered Peptone Water, Syrup
Read more -
Buffered Peptone Water, Winchester
Read more -
TSA (Tryptone Soya Agar) + Neutralisers, Contact Plate
Read more -
Baird-Parker Agar, Contact Plate
Read more -
Dey/Engley (D/E) Neutralising Agar, Contact Plate
Read more -
Rose Bengal Agar + Chloramphenicol, Contact Plate
Read more -
Columbia Blood Agar + 5% Defibrinated Horse Blood, 140mm Plate
Read more -
Tryptone Soya Agar, 55mm Plate
Read more -
Brain Heart Infusion Agar, 55mm Plate
Read more -
Malt Extract Agar, Contact Plate
Read more -
Tryptone Soya Agar (TSA) (EP/USP) Syrup
Read more -
Tryptone Soya Agar (TSA) (EP/USP) Winchester
Read more -
Tryptone Soya Agar, (TSA) (EP/USP) Universal Slope
Read more -
Streptomyces Agar, 90mm Plate
Read more -
Columbia CNA Agar + 10% Horse Blood + Colistin 10mg/l + Nalidixic Acid 10mg/l, 90mm Plate
Read more -
Fastidious Anaerobe Agar (FAA) + 5% Horse Blood, 90mm Plate
Read more -
Streptomyces Agar, 140mm Plate
Read more -
Columbia Blood Agar + 5% Defibrinated Horse Blood, Contact Plate
Read more -
Sabouraud Dextrose Agar (SDA), Contact Plate
Read more -
Tryptone Soya Agar, Contact Plate
Read more -
Mannitol Salt Agar, 90mm Plate
Read more -
Nutrient Agar, 90mm Plate
Read more -
Sabouraud Dextrose Agar, 90mm Plate
Read more -
Streptococcal Selective Agar (C.O.B.A), 90mm Plate
Read more -
Tryptone Soya Agar, 90mm Plate
Read more -
Violet Red Bile Glucose Agar (VRBGA), 90mm Plate
Read more